Recently, Dr. Wenhao Yuan from the School of Mechanical Engineering at Shanghai Jiao Tong University published a paper entitled "Elucidating the Mechanism for Oxidative Coupling of Methane Catalyzed by La2O3: Experimental and Microkinetic Modeling Studies" in ACS Catalysis. The first author is Zaili Xiong, a PhD student at the Institute of Aerospace Power from School of Mechanical Engineering, and the corresponding authors are Dr. Wenhao Yuan and Professor Fei Qi.
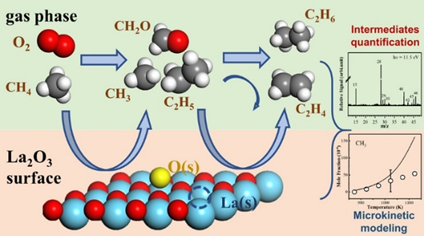
Figure 1. Schematic diagram of the micro-kinetics of OCM catalyzed by La2O3
Methane is an important C1 molecule that has attracted sustained and widespread attention in terms of its clean and efficient conversion and utilization. OCM is widely considered one of the most promising approaches for the direct conversion of methane into C2 products (such as ethane and ethylene) (Figure 1). The thermal effects of the OCM reaction may significantly influence the formation and distribution of products, where localized overheating intensifies deep oxidation, thereby reducing C2 yield. A comprehensive understanding of the complex gas-surface phase reaction mechanism of OCM and the development of microkinetic models will provide a theoretical basis for the design of catalysts and reactors, as well as the optimization of industrial operating conditions.
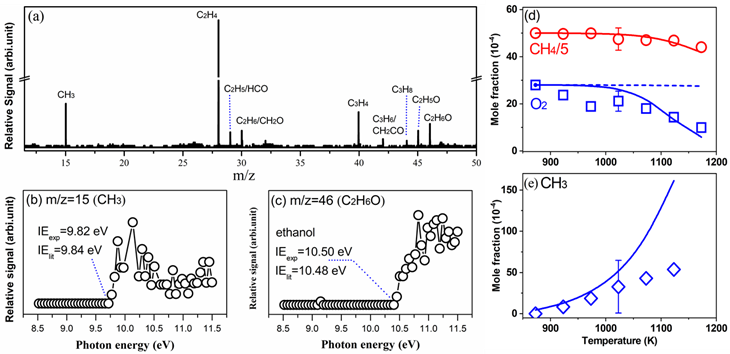
Figure 2. (a) Photoionization mass spectrometry; photoionization efficiency spectra of (b) methyl radical and (c) ethanol; comparison of experimental and simulation results of (d) reactants and (e) methyl radical.
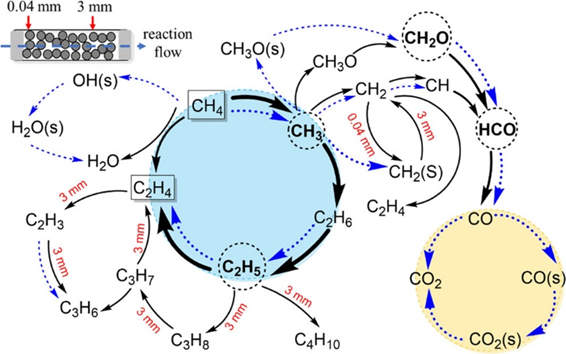
Figure 3. Reaction network of OCM catalyzed by La2O3
This work utilized synchrotron vacuum ultraviolet photoionization mass spectrometry (SVUV-PIMS) combined with a fixed-bed reactor to qualitatively and quantitatively detect intermediate species such as methyl radicals, ethyl radicals, and formaldehyde (Figures 2a-2c). The experimental results revealed the decisive role of active radicals in driving the OCM reaction process, with ethanol identified as an important C2 oxygenated intermediate formed from the reaction. In addition, this study developed a kinetic model which incorporates detailed surface and gas-phase reactions to simulate the experimental results (Figures 2d-2e) and uncover the complex reaction network of the OCM process (Figure 3). The results elucidate the contributions of gas-phase reactions and surface catalytic reactions to the OCM reaction process under different reaction temperature conditions. At lower temperatures, the OCM reaction is dominated by surface catalysis, where methane is converted through the surface reaction CH4+O(s)→CH3+OH(s); as temperature increases, the importance of gas-phase reactions gradually rises, while surface catalytic reactions still dominate. The work also clarifies the key pathways and control factors for the generation of deep oxidation products COx and C3 by-products in the OCM reaction.
The research results will provide a foundation for the optimization of La2O3-catalyzed OCM reaction systems, as well as the regulation of reaction conditions, aiming for further improvements in methane conversion rates and C2 product selectivity. The microkinetic modeling and analysis methods developed in this work can be extended to investigate various catalytic reaction systems.
This work was supported by the National Natural Science Foundation of China and the Key Research and Development Program from the Ministry of Science and Technology.
Paper Link: https://pubs.acs.org/doi/full/10.1021/acscatal.3c04714
Source: SJTU ME
Editor on Duty: Yan Cheng

